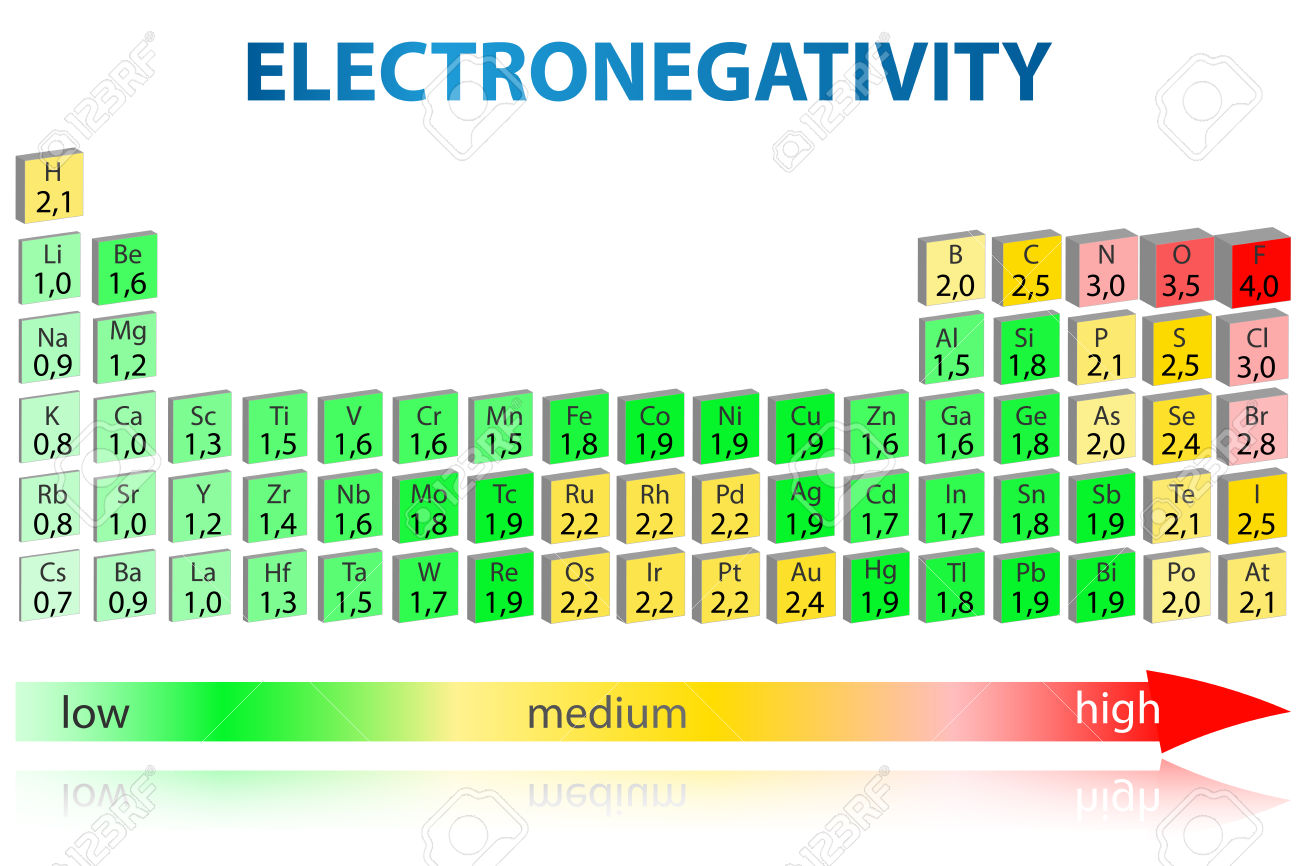
Important (and understandable) electron shifts, s→p, s→d, s→f, and d→f are essential chemical and physical consequences of compression. However, Hund’s rule is never violated for single atoms in the considered pressure range. Magnetism may increase or decrease with pressure, depending on which atom is considered. In contrast, the Madelung energy ordering rule is not predictive for atoms under compression. Our study confirms that the filling of energy levels in compressed atoms more closely follows the hydrogenic aufbau principle, where the ordering is determined by the principal quantum number. For example, we can speculate on the existence of pressure-induced polarity (red-ox) inversions in various alloys. This extension of atomic reference data assists in the working of chemical intuition at extreme pressure and can act as a guide to both experiments and computational efforts. 
Studies of 93 atoms predict drastic changes to ground-state electronic configurations and electronegativity in the pressure range of 0–300 GPa. We present a quantum mechanical model capable of describing isotropic compression of single atoms in a non-reactive neon-like environment.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
